Kusobala ukuthi umkhawulo womkhakha omusha wamandla uphakeme kunalokho obekulindelwe, njengoba imali isangena, kubonakala sengathi ifuna "i-Contemporary Amperex Technology" noma "i-BYD" elandelayo.
Ukubuka konke
Amabhethri e-sodium-ion (abizwa ngokuthi "amabhethri e-sodium") awuhlobo lwebhethri elishajekayo elisebenza ngokuhambisa ama-ion e-sodium phakathi kwe-cathode ne-anode ngesikhathi sokushaja nokukhipha. Isimiso sawo sokusebenza kanye nesakhiwo kufana namabhethri e-lithium-ion asetshenziswa kabanzi.
Kokubili i-sodium ne-lithium kungokweqembu elifanayo lezinto futhi kubonisa ukuziphatha okufanayo "kwesihlalo sokuzulazula" kokushaja nokukhipha i-electrochemical. Ngesikhathi senqubo yokushaja yebhethri le-sodium-ion, ama-sodium ion ayaphuma ku-cathode bese efakwa ku-anode kuyilapho ama-electron ehamba ngesekethe yangaphandle. Uma ama-sodium ion amaningi efakwe ku-anode, kulapho amandla okushaja aphezulu khona. Ngokuphambene nalokho, ngesikhathi sokukhipha, ama-sodium ion abuya kusuka ku-anode aye ku-cathode, okwandisa amandla okukhipha njengoba ama-sodium ion amaningi ebuyela emuva.
Isimiso Sokusebenza
Isimiso sokusebenza samabhethri e-sodium-ion sifana nesamabhethri e-lithium-ion, okuhlanganisa ukufakwa nokukhishwa kwama-ion e-sodium ukuze kufezwe ukudluliswa kweshaja. Ngesikhathi sokukhishwa, ama-ion e-sodium aphuma ezintweni ze-anode bese engena ezintweni ze-cathode, ama-electron egeleza esuka ku-anode aye ku-cathode, ekhipha amandla.
Ngesikhathi sokushaja, ama-ion e-sodium ayaphuma ezintweni ze-cathode bese ethuthela ezintweni ze-anode nge-electrolyte, kuyilapho ama-electron egeleza ezintweni ze-anode ngesekethe yangaphandle. Okungcono kakhulu, ukufakwa nokukhishwa kwama-ion ngesikhathi sokushaja nokukhipha akufanele kushintshe isakhiwo sezinto noma kubangele ukusabela okungekuhle nge-electrolyte. Kodwa-ke, ubuchwepheshe bamanje bubhekene nezinselele ngenxa yobubanzi obukhulu bama-ion e-sodium, okuholela ekushintsheni kwesakhiwo sezinto ngesikhathi sokufakwa kwama-ion, okuholela ekusebenzeni komjikelezo okwehlayo kanye nokuzinza.
Izinzuzo
Ubuningi Bamandla:Amaseli ebhethri le-sodium-ion ngokuvamile anobuningi bamandla obungu-100-150 Wh/kg, kuyilapho amaseli ebhethri le-lithium-ion ngokuvamile esukela ku-120-200 Wh/kg, kanye nezinhlelo ze-ternary ze-nickel eziphakeme ezidlula u-200 Wh/kg. Nakuba amabhethri e-sodium-ion okwamanje enobukhulu bamandla obuphansi uma kuqhathaniswa namabhethri e-ternary lithium, angahlanganisa noma amboze ububanzi bamandla obuncane bamabhethri e-lithium iron phosphate (120-200 Wh/kg) kanye namabhethri e-lead-acid (30-50 Wh/kg).
Ububanzi Bokushisa Nokuphepha Kokusebenza:Amabhethri e-sodium-ion asebenza ebangeni elibanzi lokushisa, ngokuvamile kusukela ku--40°C kuya ku-80°C. Ngokuphambene nalokho, amabhethri e-ternary lithium-ion avame ukusebenza phakathi kuka--20°C no-60°C, kanti ukusebenza kwehla ngaphansi kuka-0°C. Amabhethri e-sodium-ion angagcina isimo sokushaja esingaphezu kuka-80% (SOC) ku--20°C. Ngaphezu kwalokho, ngenxa yokumelana okuphezulu kwangaphakathi, amabhethri e-sodium-ion awathambekeli ekushiseni ngesikhathi semijikelezo emifushane, anikeza ukuphepha okukhulu uma kuqhathaniswa namabhethri e-lithium-ion.
Linganisa Ukusebenza:Ukusebenza kwezinga lokushaja kanye nokukhishwa kwamabhethri e-sodium-ion kuhlobene ngqo nekhono lokufuduka kwama-ion e-sodium esibonakalayo se-electrode-electrolyte. Izici ezithinta isivinini sokufuduka kwama-ion zithinta ukusebenza kwezinga lebhethri. Ngaphezu kwalokho, izinga lokushaja kwangaphakathi kokushisa libalulekile ekuphepheni nasekuphileni kwesikhathi eside ngesikhathi sokushaja nokukhipha izinga eliphezulu. Ngenxa yesakhiwo sawo sekristalu, amabhethri e-sodium-ion abonisa ukusebenza kahle kwezinga, okwenza afanelekele ukugcina amandla kanye nezinhlelo zokusebenza zokunikezwa kwamandla ezinkulu.
Isivinini sokushaja:Amabhethri e-sodium-ion angashajwa ngokugcwele cishe ngemizuzu eyi-10, kanti amabhethri e-ternary lithium adinga okungenani imizuzu engama-40, kanti amabhethri e-lithium iron phosphate adinga cishe imizuzu engama-45.
Ukuhlukaniswa Kwemboni
Amabhethri e-sodium-ion afika ngezinhlobo ezahlukene, okuhlanganisa amabhethri e-sodium-sulfur, amabhethri e-sodium-salt, amabhethri e-sodium-air, amabhethri e-sodium-ion aqueous, amabhethri e-sodium-ion e-organic, kanye namabhethri e-sodium-ion aqinile.
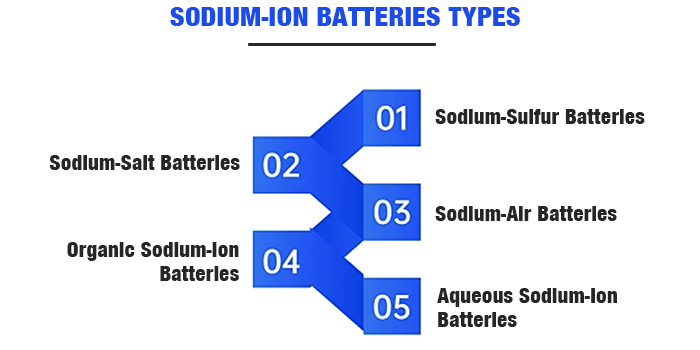
Emkhakheni wokugcina amandla, amabhethri e-sodium asetshenziswa kakhulu kwezentengiselwano afaka amabhethri e-sodium-sulfur ashisa kakhulu kanye namabhethri e-sodium-metal chloride asekelwe ezinhlelweni ze-electrolyte eziqinile. Lezi zinhlelo zisebenzisa i-sodium yensimbi njengento esebenzayo ye-anode, ebizwa ngokunembile ngokuthi amabhethri e-sodium. Ngokuvamile, igama elithi ibhethri le-sodium-ion libhekisela ezinhlotsheni ezintathu zokugcina.
Amabhethri e-Sodium-Sulfur:Lawa asebenzisa i-sodium encibilikisiwe njenge-anode kanye ne-elemental sulfur njenge-cathode, kanye ne-Al2O3 eqinile ye-ceramic njenge-electrolyte kanye ne-separator. Amabhethri e-sodium-sulfur anamandla athile aphezulu.
Amabhethri e-Sodium-Salt:Lezi zisebenzisa i-sodium ewuketshezi njenge-anode kanye nezinto ze-chloride zensimbi njenge-cathode, kanye ne-Na+ conductor Al2O3 ceramic njenge-electrolyte.
Amabhethri Omoya We-Sodium:I-cathode ngokuvamile isebenzisa izinto ezinezimbobo, ezihlinzeka ngezindlela zokusabalala kwegesi kanye nezindawo zokusabela kwe-electrode ngenxa yokumbobo kwezinto.
Amabhethri e-Organic Sodium-Ion:Lezi zisebenzisa izinto ezihlanganisiwe ze-carbon eqinile noma i-sodium ze-anode, ngezinto ze-cathode ezifaka phakathi ama-transition metal oxides kanye nama-polyanionic compounds.
Amabhethri e-Aqueous Sodium-Ion:Uma kuqhathaniswa namabhethri e-electrolyte e-organic, amabhethri e-sodium-ion asebenzisa ama-electrolyte ahlukene, anikeza ukusebenza okuphezulu kokuphepha.