Kuyabonakala ukuba umda wecandelo elitsha lamandla uphezulu kunokuba bekulindelwe, kwaye imali isangena, kubonakala ngathi ifuna i-"Contemporary Amperex Technology" okanye i-"BYD" elandelayo.
Isishwankathelo
Iibhetri zeSodium-ion (ezibizwa ngokuba "ziibhetri zeSodium") luhlobo lwebhetri etshajwayo esebenza ngokuhambisa ii-ion zeSodium phakathi kwe-cathode kunye ne-anode ngexesha lokutshaja nokukhupha. Umgaqo-nkqubo wazo wokusebenza kunye nolwakhiwo ziyafana neebhetri zeLithium-ion ezisetyenziswa kakhulu.
Zombini i-sodium kunye ne-lithium ziyinxalenye yeqela elifanayo lezinto kwaye zibonisa iindlela ezifanayo zokutshaja kunye nokukhupha i-electrochemical "chair rocking chair". Ngexesha lenkqubo yokutshaja yebhetri ye-sodium-ion, ii-sodium ion ziyaphuma kwi-cathode kwaye zingene kwi-anode ngelixa ii-electron zihamba kwisekethe yangaphandle. Okukhona ii-sodium ion zininzi kwi-anode, kokukhona amandla okutshaja aphezulu. Ngokwahlukileyo koko, ngexesha lokukhupha, ii-sodium ion zibuya kwi-anode ziye kwi-cathode, zinyusa amandla okukhupha njengoko ii-sodium ion ezininzi zibuyela umva.
Umgaqo Wokusebenza
Umgaqo osebenzayo weebhetri ze-sodium-ion ufana noweebhetri ze-lithium-ion, ezibandakanya ukufakwa kunye nokukhutshwa kwee-sodium ions ukuze kufikelelwe ekudlulisweni kwetshaja. Ngexesha lokukhupha, ii-sodium ions ziphuma kwizinto ze-anode zingene kwizinto ze-cathode, kunye nee-electron ezihamba ukusuka kwi-anode ukuya kwi-cathode, zikhupha amandla.
Ngexesha lokutshaja, ii-ion zesodiyam ziyaphuma kwizinto ze-cathode zize zingene kwizinto ze-anode nge-electrolyte, ngelixa ii-electron zingena kwizinto ze-anode ngesekethe yangaphandle. Eyona nto ifanelekileyo kukuba, ukufakwa kunye nokukhutshwa kwee-ion ngexesha lokutshaja kunye nokukhupha akufuneki kutshintshe ulwakhiwo lwezinto okanye kubangele iimpendulo ezingalindelekanga kwi-electrolyte. Nangona kunjalo, ubuchwepheshe bangoku bujongene nemingeni ngenxa yobubanzi obukhulu bee-ion zesodiyam, nto leyo ekhokelela kutshintsho lolwakhiwo lwezinto ngexesha lokufakwa kwee-ion, nto leyo ekhokelela ekuncipheni kokusebenza komjikelo kunye nokuzinza.
Iingenelo
Ubuninzi bamandla:Iiseli zebhetri zeSodium-ion zihlala zinoxinano lwamandla oluyi-100-150 Wh/kg, ngelixa iiseli zebhetri zeLithium-ion zihlala ziqala kwi-120-200 Wh/kg, kunye neenkqubo zeTerry zeNickel ezingaphaya kwe-200 Wh/kg. Nangona iibhetri zeSodium-ion okwangoku zinoxinano lwamandla oluphantsi xa kuthelekiswa neebhetri zeTerry lithium, zinokugquma okanye zigubungele uluhlu lwamandla oluphezulu lweebhetri zeLithium Iron phosphate (120-200 Wh/kg) kunye neebhetri zeLead-acid (30-50 Wh/kg).
Uluhlu lobushushu bokusebenza kunye nokhuseleko:Iibhetri zeSodium-ion zisebenza kumanqanaba obushushu abanzi, ngesiqhelo ukusuka kwi -40°C ukuya kwi 80°C. Ngokwahlukileyo koko, iibhetri ze-ternary lithium-ion zihlala zisebenza phakathi kwe -20°C kunye ne 60°C, kwaye ukusebenza kwazo kuyehla ngaphantsi kwe 0°C. Iibhetri zeSodium-ion zinokugcina ngaphezulu kwe 80% ye State of Charge (SOC) kwi -20°C. Ukongeza, ngenxa yokumelana okuphezulu kwangaphakathi, iibhetri zeSodium-ion azikwazi ukufudumeza ngexesha lee-short circuits, zibonelela ngokhuseleko olukhulu xa kuthelekiswa neebhetri zeLithium-ion.
Inqanaba lokusebenza:Ukusebenza kwesantya sokutshaja kunye nokukhupha iibhetri ze-sodium-ion kunxulumene ngokuthe ngqo nokukwazi ukufuduka kwee-sodium ions kwi-interface ye-electrode-electrolyte. Izinto ezichaphazela isantya sokufuduka kwee-ion zichaphazela ukusebenza kwesantya sebhetri. Ukongeza, izinga lokusasazeka kobushushu bangaphakathi libalulekile kukhuseleko kunye nobomi ngexesha lokutshaja nokukhupha okuphezulu. Ngenxa yesakhiwo sazo sekristale, iibhetri ze-sodium-ion zibonisa ukusebenza okuhle kwesantya, nto leyo ezenza zifaneleke ukugcina amandla kunye nokusetyenziswa kombane omkhulu.
Isantya sokutshaja:Iibhetri zeSodium-ion zinokutshajwa ngokupheleleyo kwimizuzu eli-10, ngelixa iibhetri ze-ternary lithium zifuna ubuncinane imizuzu engama-40, kwaye iibhetri ze-lithium iron phosphate zifuna malunga nemizuzu engama-45.
Ulwahlulo lweShishini
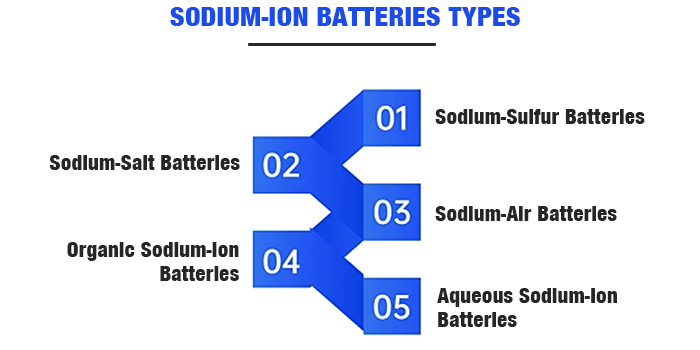
Iibhetri zeSodium-ion ziza ngeentlobo ezahlukeneyo, kuquka iibhetri zeSodium-sulfur, iibhetri zeSodium-salt, iibhetri zeSodium-air, iibhetri zeSodium-ion zamanzi, iibhetri zeSodium-ion zeOrganic, kunye neebhetri zeSodium-ion zeSodium-state eziqinileyo.
Kwicandelo lokugcina amandla, iibhetri zesodium ezisetyenziswa kakhulu kurhwebo ziquka iibhetri zesodium-sulfur ezishushu kakhulu kunye neebhetri zesodium-metal chloride ezisekelwe kwiinkqubo ze-electrolyte eziqinileyo. Ezi nkqubo zisebenzisa i-metallic sodium njengezinto ezisebenzayo ze-anode, ezibizwa ngokuchanekileyo ngokuba ziibhetri zesodium. Ngokwesiqhelo, igama elithi sodium-ion battery libhekisa kwiindidi ezintathu zokugqibela.
Iibhetri zeSodium-Sulfur:Ezi zisebenzisa i-sodium enyibilikisiweyo njenge-anode kunye ne-elemental sulfur njenge-cathode, kunye ne-Al2O3 eqinileyo ye-ceramic njenge-electrolyte kunye ne-separator. Iibhetri ze-sodium-sulfur zinamandla athile aphezulu.
Iibhetri zeSodium-Salt:Ezi zisebenzisa i-sodium engamanzi njenge-anode kunye nezinto ze-chloride zesinyithi njenge-cathode, kunye ne-Na+ conductor Al2O3 ceramic njenge-electrolyte.
Iibhetri zoMoya zeSodium:I-cathode isebenzisa izinto ezinemibhobho, ezibonelela ngeendlela zokusasazwa kwegesi kunye neendawo zokusabela kwe-electrode ngenxa yokuvuleka kwezinto.
Iibhetri ze-Organic Sodium-Ion:Ezi zisebenzisa izinto ezidityaniswe nekhabhoni eqinileyo okanye i-sodium kwi-anode, kunye nezinto ze-cathode kuquka ii-transition metal oxides kunye nee-polyanionic compounds.
Iibhetri zeSodium-Ion ezingamanzi:Xa kuthelekiswa neebhetri ze-electrolyte zendalo, iibhetri ze-sodium-ion zamanzi zisebenzisa ii-electrolyte ezahlukeneyo, zibonelela ngokusebenza okuphezulu kokhuseleko.